Advancing Aortic Technologies
with Purpose™







May 18, 2026
Artivion Completes Acquisition of Endospan Ltd.
Today, we announced that we have completed the acquisition of our long-standing partner Endospan Ltd. (“Endospan”), a pioneer in the endovascular repair of aortic arch disease and developer of the NEXUS® Aortic Arch System.
January 27, 2025
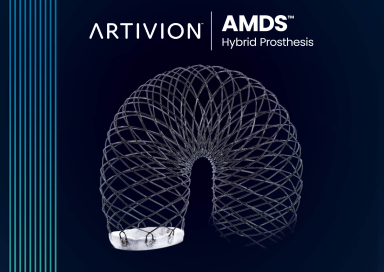
Late-Breaking Data from AMDS PERSEVERE Trial at the 61stSociety of Thoracic Surgery Annual Meeting
Today we announced data from the AMDS PERSEVERE clinical trial (the “IDE”) was presented in a Late-Breaking Science presentation at the 61st Annual Meeting of the Society of Thoracic Surgeons in Los Angeles, California. The data presented analyzed clinical outcomes across the full 93 study participant IDE cohort at one year following AMDS implantation..

December 9, 2024
Artivion Granted FDA Humanitarian Device Exemption for the AMDS Hybrid Prosthesis
Today, Artivion announced the U.S. Food and Drug Administration (FDA) has granted a Humanitarian Device Exemption (HDE) for the use of the AMDS Hybrid Prosthesis (“AMDS”) in acute DeBakey Type I dissections in the presence of malperfusion. The AMDS is the world’s first aortic arch remodeling device for use in treating acute DeBakey Type I aortic dissections.

November 9, 2023
Artivion Announces Completion of Enrollment in PERSEVERE Trial.
The PERSEVERE trial is a prospective, multicenter, non-randomized clinical trial to determine if patients with acute DeBakey Type I aortic dissection can be treated safely and effectively using the AMDS Hybrid Prosthesis.
When the need is aortic,
the solution is Artivion
Our intentional focus on the aorta and collaboration with the world’s foremost cardiac and vascular surgeons allow us to leverage our combined expertise in the development of new, innovative, life-changing aortic-centric technologies.
Please contact your local Artivion representative for details.
HEART VALVE
SOLUTIONS

ALLOGRAFTS
CARDIAC & VASCULAR

- CRYOVALVE® SG PULMONARY HUMAN HEART VALVE
- CRYOVALVE® AORTIC ALLOGRAFT
- CRYOPATCH® SG PULMONARY PATCH
- CRYOGRAFT® DESCENDING THORACIC AORTA
- CRYOARTERY® AORTOILIAC ARTERY
- CRYOARTERY® FEMORAL ARTERY
- CRYOVEIN® SAPHENOUS VEIN
- CRYOVEIN® FEMORAL VEIN
- CRYOVEIN® PC PEDIATRIC CONDUIT
- CRYOPATCH® AUTOLOGOUS PERICARDIUM
AORTIC ARCH
SOLUTIONS

THORACOABDOMINAL
SOLUTIONS

ANCILLARY SOLUTIONS
CARDIAC & VASCULAR

E-XTRA DESIGN ENGINEERING

Patient-Powered Progress
We provide solutions that have far-reaching impact on surgical outcomes and restoring the lives of patients fighting aortic diseases. Cardiac and vascular surgeons the world over—along with each patient in their care—have greatly benefited from our life-changing technologies.